APIs
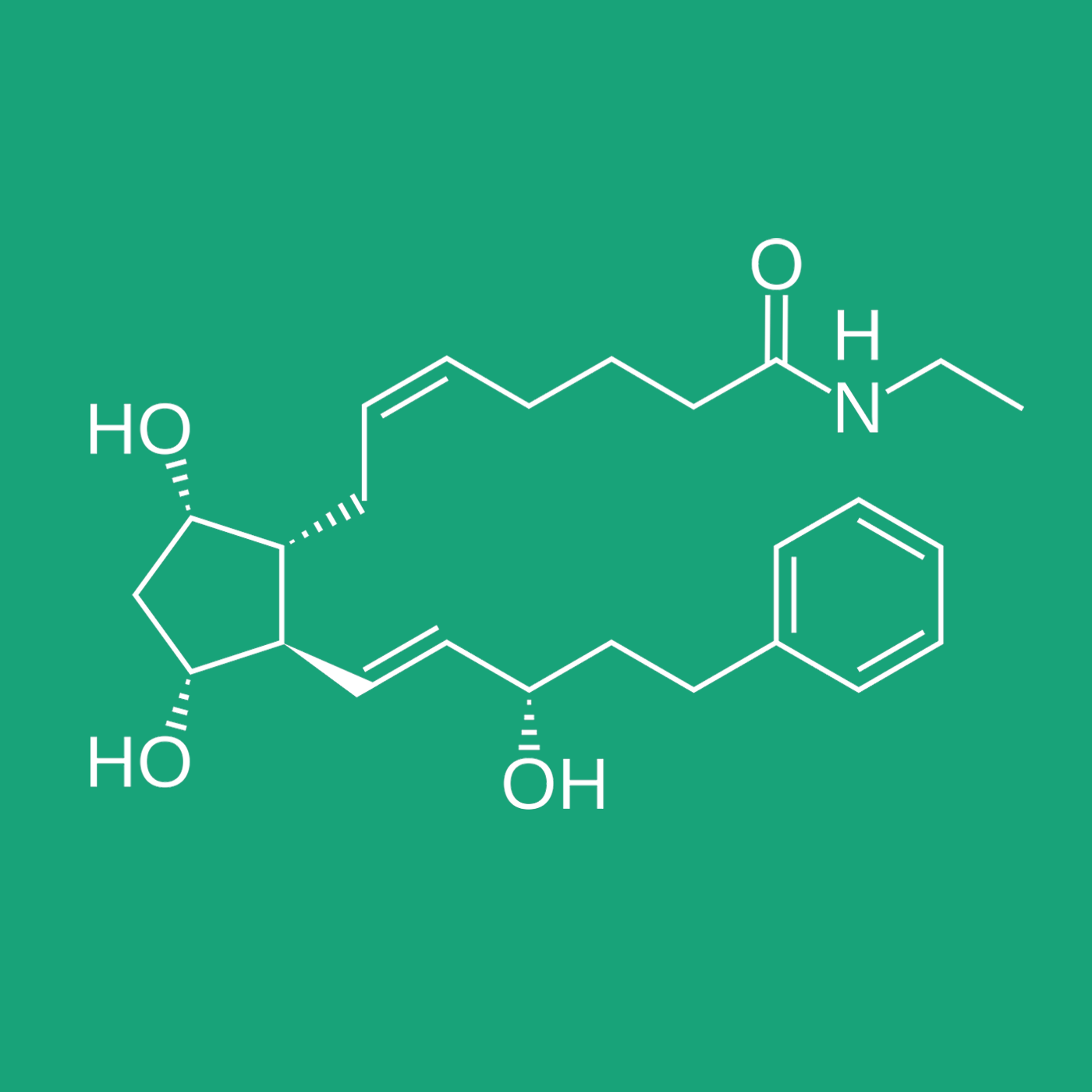
BIMATOPROST
Bimatoprost, a hypotensive agent, is a synthetic amide analogue (prostamide) of the potent FP receptor agonist 17-phenyl-prostaglandin F2α (PGF2α). Bimatoprost was first reported in the early 2000s and marketed, among others, as LumiganTM.1 It is approved as the first-line treatment for glaucoma in the European Glaucoma Society guidelines.2
C2 PHARMA is one of the leading manufacturers and suppliers of Bimatoprost.
USES
As 0.03% ophthalmic solution it is approved for treatment of ocular hypertension and glaucoma as well as for hypotrichosis of eyelashes.
MECHANISM OF ACTION
Although classified as a prostamide, Bimatoprost exhibits a similar mechanism of action as the prostaglandins. It lowers the IOP without significantly affecting the aqueous production rate and enhances the aqueous humor outflow, either by enhancing the pressure-sensitive (presumed trabecular) outflow pathway or by increasing the pressure-insensitive (uveoscleral) outflow.3
REFERENCES
- Woodward, D. F. Survey of Ophthalmology, 2001, 45, 337-345.
- European Glaucoma Society. Terminology and Guidelines for Glaucoma. 3rd ed. Savona: Dogma; 2008.
- Sheng Lim, K. et al. Ophthalmology, 2008, 115, 790-795.